How Do You Know When the Sample Is Too Concentration or Dilute for an Absorption Spectrum

Theoretical principles
Introduction
Many molecules absorb ultraviolet or visible light. The absorbance of a solution increases equally attenuation of the beam increases. Absorbance is directly proportional to the path length, b, and the concentration, c, of the absorbing species. Beer's Law states thatA = ebc , where e is a constant of proportionality, called the absorbtivity.
Different molecules absorb radiation of different wavelengths. An assimilation spectrum will show a number of assimilation bands respective to structural groups within the molecule. For instance, the absorption that is observed in the UV region for the carbonyl group in acetone is of the same wavelength equally the absorption from the carbonyl grouping in diethyl ketone.
For a comprehensive word of Beer's Police force, click here
Electronic transitions
The assimilation of UV or visible radiation corresponds to the excitation of outer electrons. There are three types of electronic transition which can exist considered;- Transitions involving p, south, and due north electrons
- Transitions involving accuse-transfer electrons
- Transitions involving d and f electrons (non covered in this Unit)
When an atom or molecule absorbs energy, electrons are promoted from their footing land to an excited state. In a molecule, the atoms can rotate and vibrate with respect to each other. These vibrations and rotations as well have discrete energy levels, which can be considered as being packed on superlative of each electronic level.

Absorbing species containing p, south, and n electrons
Assimilation of ultraviolet and visible radiation in organic molecules is restricted to sure functional groups (chromophores) that contain valence electrons of low excitation free energy. The spectrum of a molecule containing these chromophores is circuitous. This is considering the superposition of rotational and vibrational transitions on the electronic transitions gives a combination of overlapping lines. This appears as a continuous absorption band.Possible electronic transitions of p, s, and n electrons are;
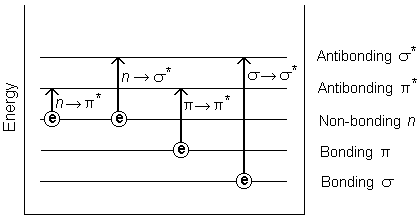
s ® due south * Transitions
An electron in a bonding due south orbital is excited to the corresponding antibonding orbital. The energy required is large. For example, methyl hydride (which has only C-H bonds, and can only undergo s ® s * transitions) shows an absorbance maximum at 125 nm. Assimilation maxima due to southward ® s * transitions are not seen in typical UV-Vis. spectra (200 - 700 nm)
n ® s * Transitions
Saturated compounds containing atoms with lone pairs (not-bonding electrons) are capable of due north ® s * transitions. These transitions commonly need less free energy than s ® due south * transitions. They tin be initiated by light whose wavelength is in the range 150 - 250 nm. The number of organic functional groups with n ® s * peaks in the UV region is small.
n ® p * and p ® p * Transitions
Almost absorption spectroscopy of organic compounds is based on transitions of n or p electrons to the p * excited state. This is because the absorption peaks for these transitions fall in an experimentally convenient region of the spectrum (200 - 700 nm). These transitions need an unsaturated group in the molecule to provide the p electrons.
Molar absorbtivities from n ® p * transitions are relatively low, and range from 10 to100 50 mol-one cm-1 . p ® p * transitions normally give molar absorbtivities between 1000 and ten,000 50 mol-1 cm-ane .
The solvent in which the absorbing species is dissolved also has an effect on the spectrum of the species. Peaks resulting from n ® p * transitions are shifted to shorter wavelengths (blue shift) with increasing solvent polarity. This arises from increased solvation of the solitary pair, which lowers the energy of the n orbital. Oft (just not e'er), the reverse (i.e. ruby shift) is seen for p ® p * transitions. This is caused past attractive polarisation forces betwixt the solvent and the absorber, which lower the energy levels of both the excited and unexcited states. This effect is greater for the excited state, and and so the free energy difference between the excited and unexcited states is slightly reduced - resulting in a small ruby-red shift. This effect as well influences n ® p * transitions but is overshadowed by the blue shift resulting from solvation of lone pairs.
Accuse - Transfer Absorption
Many inorganic species prove accuse-transfer absorption and are called accuse-transfer complexes. For a circuitous to demonstrate charge-transfer behaviour, one of its components must have electron donating backdrop and another component must be able to have electrons. Absorption of radiation so involves the transfer of an electron from the donor to an orbital associated with the acceptor.
Molar absorbtivities from charge-transfer assimilation are big (greater that 10,000 Fifty mol-ane cm-1).
Review your learning
You should now exist aware of why molecules absorb radiation in the UV and visible low-cal regions, and why absorption spectra look the way they do.
Subsequently a short quiz, nosotros volition be because the practical aspects of UV - Vis. spectroscopy, and looking at the instrumentation needed to perform this technique.
|
 Biosciences Homepage
Biosciences Homepage 
Source: https://teaching.shu.ac.uk/hwb/chemistry/tutorials/molspec/uvvisab1.htm

0 Response to "How Do You Know When the Sample Is Too Concentration or Dilute for an Absorption Spectrum"
Post a Comment